|
The Wisconsin Department of Natural Resources regulates the amount of chromium that can be released by industries. No standards exist for the amount of chromium allowed in the air of homes however, occupational standards apply to industries where exposure to chromium may occur. What regulations and guidelines are available to protect people from chromium Air Chromium can pass through the skin, but this is probably not a major route of exposure. Exposure can be reduced by thorough washing of exposed skin and clothing to remove soil residues. Contact with contaminated soils can result in exposure to chromium. Plants can absorb chromium, which can then be passed to those who eat the plants. Most human exposure to chromium through drinking and eating comes in the form of trivalent chromium found in fresh vegetables, meats, fish, and poultry.ĭrinking water is not normally a major source of exposure. This is the route of exposure that is of greatest concern. People can be exposed to chromium by breathing chromium dust or fumes. Many chromium-containing compounds are used for plating, manufacturing paints and dyes, tanning leather and preserving wood. Metallic chromium is mined for use in steel and other metal products. It is the most toxic form of chromium, and has been shown to cause lung cancer when workers are exposed to high air levels for long time periods. "Hexavalent" chromium can occur naturally, but can also be produced by certain industrial processes. The less-toxic forms of chromium are used to make flooring materials, video and audio recording tapes, stainless steel, chrome-plated items and copy machine toner. The normal intake from eating foods that are high in natural chromium is 70-80 micrograms per day and is considered safe. "Trivalent" chromium is naturally occurring and is essential for good health. This permits a method detection limit (MDL) for chromate of 0.02 μg/L and can support a reporting limit of 0.06 μg/L.Learn what you need to know about chromium.Ĭhromium is a naturally occurring element which can be primarily be found in two different forms: "trivalent" chromium and "hexavalent" chromium. This method is based on anion-exchange chromatography and detection after postcolumn reaction with 1,5-Diphenylcarbazide (CAS RN 140‑22‑7), which yields a compound with visible absorbance at 530 nm. Based on this review, OEHHA in the state of California recently issued a new PHG for chromate at 0.02 μg/L in drinking water.Ĭurrently, dissolved hexavalent chromium is measured according to a modified version of US EPA Method 218.6. In September 2010, the EPA released the Toxicological Review of Hexavalent Chromium. 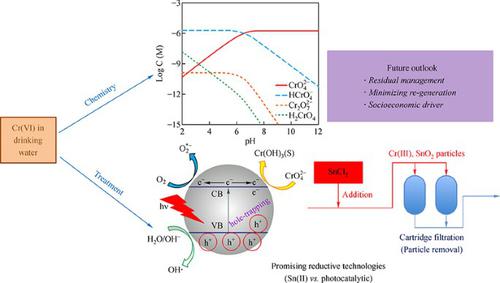 
In 2009, the Office of Environmental Health Hazard Assessment (OEHHA) at the California EPA proposed to lower the PHG for Cr(VI) to 0.06 μg/L. In 2008, the agency conducted a comprehensive review of the health effects of chromate based on toxicity studies done by the US National Toxicology Program. The PHG is based on an estimated one-in-one-million lifetime cancer risk level.ĭrinking water standards are regularly re‑evaluated by the US Environmental Protection Agency (EPA). For example, in 1999, the state of California established a public health goal (PHG) of 0.2 μg/L for Cr(VI) and 2.5 μg/L for total chromium. Hence, chromates are regulated in the environment and are a primary drinking water contaminant in the United States. All hexavalent chromium Cr(VI) compounds are strong oxidizing agents and considered toxic and potentially carcinogenic. Chromates are oxyanions of chromium in oxidation state +6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
